Aug
04
2021
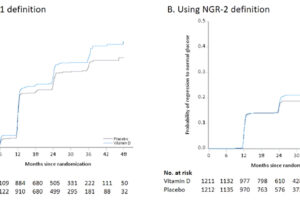
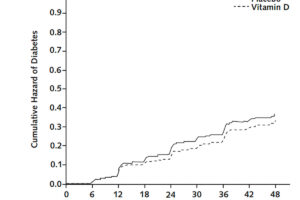
Despite evidence for associations between low circulating 25(OH)D concentration and kidney disease, few clinical trials have evaluated the effect of vitamin D supplementation on kidney outcomes. The Vitamin D and Type 2 Diabetes (D2d) study was a randomized clinical...










 Know someone, a friend or family member, who might be interested in learning about D2d? You can easily share with others by email or on social media.
Know someone, a friend or family member, who might be interested in learning about D2d? You can easily share with others by email or on social media.